Joan Camarro Simard, Department Editor
Case Study from the December 1999 issue of the ANNA Journal
Hazel A. Dennison
Patient Profile
W.T., a 43-year-old male, began on hemodialysis in 1996 for membranoproliferative glomerulonephritis (MPGN). His height was 5’10” and his weight was 330 lbs. He supported his wife and 3 children by working as a security guard. He accepted his diagnosis and the need for his hemodialysis treatments while awaiting a cadaver kidney transplant.
A polytetrafluorethylene (PTFE) graft was placed to begin hemodialysis. Blood urea recirculation data revealed a rate of less than 5%. His initial treatment regimen included Baxter CA 210 cellulose acetate dialyzer (Baxter Health Care, [Kuf of 10.1/surface area of 2.1 ml]), a blood flow of 400 cc/minute, a dialysate flow of 600 cc/minute, and a 4-hour treatment regimen. Lab data drawn after 2 months on dialysis revealed a hemoglobin (Hgb) of 11.4 g/dl; transferrin saturation (Tsat) of 28%; a blood urea nitrogen recirculation study of 4%, and a urea reduction ratio (URR) of .48. W.T. continued to complain of fatigue, nausea, and ineffective sleep patterns.
Nursing Diagnosis
Our goal was to assist W.T. to regain his former state of health. In doing so, we obtained our list of patient problems: (a) ineffective sleep patterns related to reduced URR; (b) alteration in activity level—decreased activity related to ineffective dialysis; and (c) nausea related to ineffective dialysis and reduced URR.
Discussion
During the next several months, treatment changes included using a larger filter, a BK2.1U polymethyl methacrylate membrane (Toray, [Kuf 19 surface area 2.1]), and increased blood flows up to 500 cc/minute. These interventions resulted in minimal improvement in the URR to .50. Patient education with W.T. became a priority. Members of the multidisciplinary team discussed the adequacy of dialysis, protein intake, and weight reduction. Our focused approach improved the URR from .50 to .52. This continued to be less than our acceptable goal of a minimum of .65.
We requested that W.T. allow us to change his dialyzer mid-treatment to evaluate the amount of protein binding to the dialyzer membrane (Ringoir & Vanholder, 1992). Although it was theorized that this could account for the decrease in dialyzer clearance, there was no appreciable difference in his total URR (.51). We then requested that W.T. stay for 2 hours after dialysis to evaluate his amount of urea rebound (Kerr, Argiles, & Canaud, 1993; Starr, 1992). As suspected, the amount of rebound was discernible at 13%. The vascular access was again evaluated. Venous pressures measured during the first 2-5 minutes of dialysis at a 200 cc/min blood flow were found acceptable in the 100-120 cc/min range with 15 gauge hemodialysis needles (Beathard, 1992). The blood urea recirculation rate remained stable at 5%.
The next intervention attempted was to increase W.T.’s dialysis time to 5-hour treatments. The healthcare team spoke with W.T., and he was amenable to trying this approach. However, he often could not remain on dialysis for the full 5-hour treatments due to time constraints from his job and family obligations. At this point, W.T. remained on a BK2.1U (Toray, [Kuf 19 and surface area 2.1]) with a blood flow rate of 500 cc/minute, a dialysate flow rate of 600 cc/minute for 5 hours, and a URR of .56.
A conference entitled “The Future of Dialysis Modalities,” sponsored by Baxter Renal Care, presented “The Use of In-Series Dialyzers” as a method of improving URR (Hansen, 1997). We regained enthusiasm for our approach to W.T.’s treatment. A team effort was paramount. The idea was conceptualized and modified into our facility’s procedures.
First, the nurse educator obtained the needed connector adapter. The biomedical department assisted in obtaining a dialysate flow line. A “dry run” (without patient involvement) consisting of the adapters, lines, and flow paths all worked well together. The medical director and physicians viewed the results of the work-up and agreed to a trial run on an appropriate patient. The nurse educator presented the project to the staff and developed a sample model of the system to assist with set-up. A technician developed a drawing that showed the connections and flow paths of the blood and dialysate.
The clinical nurse specialist approached W.T. with the idea. He was willing to attempt in-series dialyzers if it meant that he could obtain adequate treatment without further increase in his treatment time. We requested that he remain on 5-hour treatments so we could compare the in-series treatment URR against previous results.
Initially, we used two Alwall GFS+20 (Gambro Health Care, [Kuf 11.4 and a surface area of 1.8]) dialyzers in series. Our trial revealed a 41% clearance of the first dialyzer (preBUN 49 mg/dl[A1]/post BUN 29[V1]) and a 62% clearance of the second dialyzer (preBUN 29[V1]/post BUN 11[V2]). This resulted in an overall clearance of 78% (1 – V2/A1 [see Figure 1]). In the first month of in-series use, we obtained a URR on W.T. of .64. In the several months after our initial triumph, W.T.’s condition began to improve. His nausea and sleep disturbances subsided and his energy returned resulting in an increase in activity. W.T. was transplanted in November 1998, 6 months after in-series dialysis, and continues to do well.
The physician, the clinical nurse specialist, the nurse educator, the biomedical engineer, the dietitian, the staff nurses, and the technicians worked as a whole to provide successful results. Each team member had individual knowledge, experience, and skills that were integrated into a strategy to improve patient outcomes. This case revealed a team effort and approach to problem-solving.
Summary
In larger clients where standard methods of urea removal do not provide adequate dialysis, the increase in surface area of in-series dialyzers has accomplished this goal. Each step that we took imparted to W.T. that our concern was to improve his health.
Our objective was to improve the URR in W.T. when his rigorous “standard” treatment did not provide adequate dialysis. An in-series set of dialyzers was designed and configured, necessary supplies were obtained, staff education was performed, and a pilot study was attempted with W.T. An improvement in URR from .52 to .64 occurred during the first month of in-series dialyzers. This resulted in an increased urea reduction of 23%. Once we recognized the usefulness of in-series dialyzers as a tool to improve URR, we applied it to several other patients with similar success rates. The benefits of in-series dialyzers include cost-effectiveness (we realized a cost savings of $6 per treatment), higher URR capabilities, the ability to provide adequate dialysis to outliers, improved compliance with treatment times, and a method to maintain a healthy client.
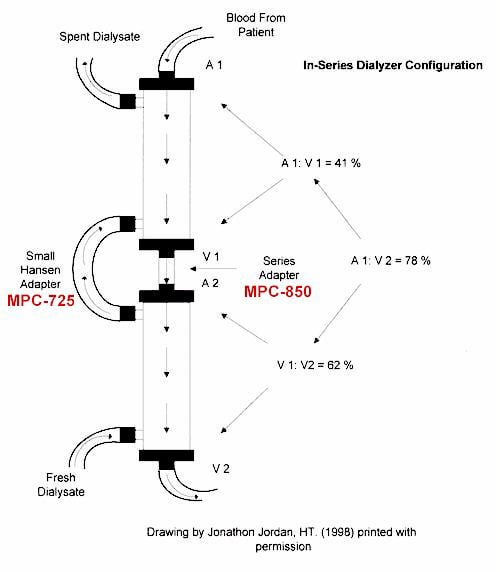
Note: Drawing by Jonathan Jordan, HT. (1998) printed with permission.
Labeled area of Blood Sample –
A1 = Sample of BUN from arterial blood line that connects to dialyzer #1.
V1 (or A2) = Sample of BUN from venous blood line adapter that connects dialyzer #1 and #2.
V2 = Sample of BUN from venous blood line after it has cleared through dialyzer #2.
The Case Study department of the ANNA Journal invites nephrology nurses of all levels and subspecialties to share their clinical experiences with their colleagues. The purpose of a Case Study is to educate others through discussion of a patient’s clinical presentation and nursing interventions. Practitioners and educators are encouraged to submit case studies that address their patient-related nursing care and solutions to situations encountered in the care of a patient with renal disease and/or in performing extracorporeal therapies. The author guidelines, which appear in the February issue each year, are a suggested format intended to assist the novice writer who needs assistance with the organization of material. Please contact Joan Camarro Simard, department editor, through the ANNA National Office for assistance or with questions. Submit a final copy to the ANNA Journal; ANNA National Office; East Holly Avenue Box 56; Pitman, NJ 08071-0056; telephone (856) 256-2320; fax (856) 589-7463. The opinions and assertions contained herein are the private views of the contributors and do not necessarily reflect the views of the American Nephrology Nurses’ Association.
| References Beathard, G.A. ( 1992). Physical examination of av grafts. Seminars in Dialysis, 5, 74. |
Hazel A. Dennison, MSN, RN, CNN, CS, is a clinical nurse specialist, at Kennedy Dialysis Centers in Stratford and Washington Township, NJ, and is an ANNA Continuing Education Approver Board member and an ANNA Journal manuscript reviewer. |
